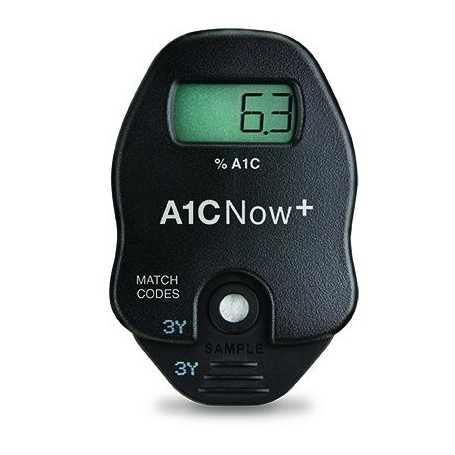
This innovative technology enables you to communicate face-to-face with your patients about their diabetes control and easily measure Glycated Hemoglobin
- More efficient than the lab
- A1CNow+ provides results in 5 minutes
- Small (5μL) blood sample
- A1CNow+ is portable and can be used in multiple exam rooms
- Easy to use – minimal training required
- No maintenance is required by A1CNow+
- Lab accurate
- National Glycohemoglobin Standardization Program certified
- CLIA waived
- Reimbursable
You will receive 1 analyzer to use with the 10 test strips included. Once you have used the 10 strips, you will have to throw away the analyzer included in the package. Each kit will come with an analyzer that is specifically coded for the number of test strips included with it.
What are the advantages of the A1CNow+ test by Chek Diagnostics?
A1CNow+ is fast, easy, and accurate. It provides A1C results in 5 minutes which are 99% laboratory accurate*. These A1C tests are for use in a doctors office. Providing real-time results outside of the laboratory, A1CNow+ is a hand-held, portable monitor which allows A1C testing in every exam room. Real-time A1C results allow for timely decisions on therapy changes when needed. The product requires no maintenance. Office staff no longer have to prepare and refrigerate lab samples. Obtaining A1C results during the patient office visit eliminates follow-up phone calls to discuss results and possible therapeutic changes. Our A1CNow® System is CLIA waived and certified by the National Glycohemoglobin Standardization Program (NGSP).
* Study results with healthcare professionals showed that the accuracy of A1CNow+ with fingerstick samples was, on average, 99%. This means that, on average, a true 7.0% A1C could read approximately 6.9%A1C. An individual A1CNow+ result may differ by as much as -1.0% A1C to +0.8% A1C from the true result. This represents the 95% confidence limits of a Bland-Altman plot.
How does the A1CNow+ test work?
The single-use test provides quantitative results from a single drop of blood. Lance the finger for an adequate drop of blood. Touch the blood drop with the tip of the blood collector. Insert the blood collector into the open end of the sampler body and shake. Insert the cartridge into the A1CNow+ monitor. Deliver the sampler body into the cartridge. Your result will display after 5 minutes. Results are 99% laboratory accurate*.
*Study results with healthcare professionals showed that the accuracy of A1CNow+ with fingerstick samples was, on average, 99%. This means that, on average, a true 7.0% A1C could read approximately 6.9%A1C. An individual A1CNow+ result may differ by as much as -1.0% A1C to +0.8% A1C from the true result. This represents the 95% confidence limits of a Bland-Altman plot
What is the National Glycohemoglobin Standardization Program (NGSP)?
The NGSP standardizes glycated hemoglobin test results so that clinical laboratory results are comparable to those reported in the Diabetes Control and Complications Trial (DCCT) where relationships to mean blood glucose and risk for vascular complications have been established. A key component of the program is the Reference Laboratory Network. The network interacts with manufacturers of glycohemoglobin methods to assist them first in standardizing their methods and then in providing comparison data for certification of traceability to the DCCT.
What is the Clinical Laboratory Improvements Amendment (CLIA)?
Clinical Laboratory Improvements Amendment (CLIA) is an act of Congress that established quality standards for all laboratory testing to ensure the accuracy, reliability, and timeliness of patient test results regardless of where the test was performed.
What does CLIA waived mean?
A CLIA waived test has been reviewed by the Food & Drug Administration (FDA) and Centers for Medicare and Medicaid Services (CMS). It is defined as simple laboratory examinations and procedures that are cleared by the FDA for home use; employ methodologies that are so simple and accurate as to render the likelihood of erroneous results negligible; or pose no reasonable risk of harm to the patient if the test is performed incorrectly.
Data sheet
- Factor Analysis
- glycosylated hemoglobin
- Type
- Blood analyzer